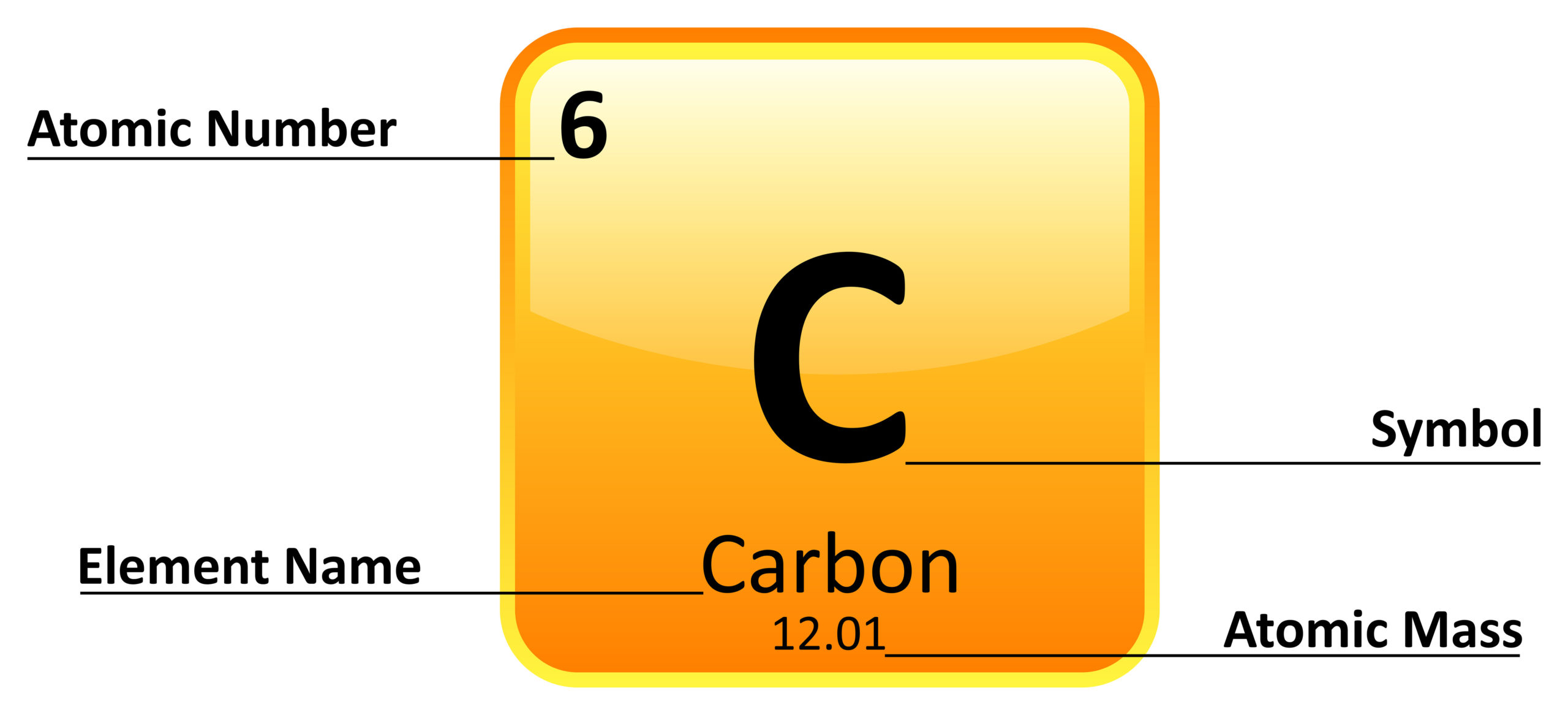
Scientists created a variety of artificial elements. This analogy piqued people’s interest in the chemistry of these elements. Chemists discovered an analogy between the 94 naturally occurring chemical elements after learning about the periodic law. Scientists now had a clear understanding of quantum numbers and the electronic configuration of elements in the periodic table after learning about the fundamental unit of elements. The number of electrons or protons in a neutral atom equals the atomic number. The modern periodic law states-” The physical and chemical properties of the elements are the periodic functions of their atomic numbers”. Dmitri Mendeleev (Source: Wikimedia) Modern Periodic Law Despite the fact that some of his predictions were incorrect, he had enough hits to establish his table as the foundation for our understanding of the elements and to confirm his place as one of the founders of modern chemistry. Mendeleev left out important elements such as noble gases because not all of the elements had been discovered at the time of his publication There were some stumbling blocks on the table. Germanium, gallium, and scandium were among these elements. Mendeleev’s table was so accurate that he could predict elements that were unknown to him at the time. Chemical properties were similar when the elements were ordered by their atomic weights. His table arranged the elements according to their atomic weights (molar masses). Mendeleev was a Russian-born chemist who was the first to publish a modern periodic table. Lothar Meyer had independently reached a similar conclusion, which he published after Mendeleyev’s paper was published.įather of Periodic Table: Dmitri Mendeleevĭmitri Mendeleev was the scientist who brought it all together (1834 to 1907). Mendeleyev in 1869 as a result of an extensive correlation of the properties and the atomic weights of the elements, with special attention to valency (that is, the number of single bonds the element can form) stated, that the periodic law “the elements arranged according to the magnitude of atomic weights show a periodic change of properties”. Newlands proposed classifying the elements in increasing atomic weight order in 1864, with the elements assigned ordinal numbers from unity upward and divided into seven groups with properties closely related to the first seven elements known at the time: hydrogen, lithium, beryllium, boron, carbon, nitrogen, and oxygen. These versions, however, were relatively simple, somewhat obscure, and difficult to read. Scientists such as John Newlands and Alexandre-Emile Béguyer de Chancourtois created periodic tables. At the same time, oxygen, sulfur, selenium, and tellurium were classified as one family. Cooke expanded Döbereiner’s suggestions by demonstrating that similar relationships extended beyond the triads of elements, with fluorine added to the halogens and magnesium added to the alkaline-earth metals. Döbereiner demonstrated in 1817 that the combining weight, or atomic weight, of strontium, is midway between those of calcium and barium, and he later demonstrated that other such “triads” exist (chlorine, bromine, and iodine, and lithium, sodium, and potassium ).īetween 18, J.-B.-A. Contributors in the development of Periodic Table This rapid expansion of chemical knowledge soon necessitated classification for the systematized literature of chemistry but also the laboratory disciplines by which chemistry is passed down from one generation of chemists to the next. The art of differentiating between chemical substances underwent rapid development in the early 19th century, which led to the accumulation of a vast body of knowledge about the chemical and physical characteristics of both elements and compounds. It’s as common to see chemical elements arranged in the modern periodic table as it is to see a world map, but it wasn’t always that way. The first 94 elements of the periodic table occur naturally, while the remaining elements from 95 to 118 have only been synthesized in laboratories or nuclear reactors. 
As a result, as the atom’s energy level rises, so does the number of energy sub-levels per energy level. The Elements with the same valence electron configuration will have similar chemical properties.Įlements in the same period, on the other hand, will have an increasing order of valence electrons. Elements are arranged in increasing atomic number order from left to right and top to bottom. They are organized in a tabular format, where a row represents a period, and a column represents a group. The periodic table is an arrangement of all known elements in order of increasing atomic number and recurring chemical properties. Importance/ Application of Periodic Table.Contributors in the development of Periodic Table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
